Despite how essential plants are for life on Earth, little is known about how parts of plant cells orchestrate growth and greening. By creating mutant plants, UC Riverside researchers have uncovered a cellular communication pathway sought by scientists for decades.
Both plants and humans have specialized light-sensitive proteins. In humans these proteins reside in the retina, allowing us to see. In plants, they are called phytochromes and are housed mainly in the nucleus, which serves as master control for the cell’s activities.
The process of photosynthesis, which converts carbon dioxide into sugar and fuels plant growth, begins when light hits the phytochromes in the nucleus. The nucleus then has to send a command to a sub-organ called a plastid to transform itself into a chloroplast, which manufactures the green pigment chlorophyll.
“The nucleus is like the federal government of the cell, while a sub-organ called the plastid functions more like the state,” said UCR’s Meng Chen, an associate professor of cell biology whose lab is one of few in the world focused on phytochrome communications. “Until now, we did not know how the nucleus sent the ‘turn green’ command to the plastids, telling them to activate their photosynthesis genes.”
The way Chen’s team arrived at the answer is detailed in two new papers published today in the journal Nature Communications.
Historically, part of the challenge has been identifying which of the 25,000 nuclear genes is responsible for regulating the cell’s greening process. To find the regulators, Chen and his team reasoned that the same genes must control not only plant greening, but other processes as well, such as height.
“The regulator we were looking for would control both qualities, height and color,” Chen said.
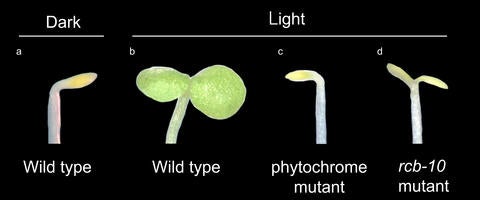
They took a small flowering plant and chemically created versions of it unable to manufacture chloroplasts, even when exposed to light. Next, they looked for mutants that are both albino and tall. As luck would have it, Chen’s team found they’d created some mutants with both qualities.
Comparing the wild plant DNA with the mutated plant DNA allowed the team to identify two genes responsible for regulating greening.
“Plants without either of these genes fail to respond to light, becoming tall and albino seedlings,” said study co-author Chan Yul Yoo, a UCR molecular biologist and first author of both papers.
Understanding the master control of chloroplast development could have profound implications for new technologies to improve crop yields and help plants cope with climate change. But the benefits of this discovery are not limited to plants. Chen’s laboratory is funded by the National Institutes of Health because of the implications of this work on cancer research.
Mitochondria, the power generators of plant and animal cells, play a role in cancer because they are involved in programmed cell death. Communications between a cell’s nucleus and mitochondria are analogous to communications between a plant cell nucleus and chloroplasts.
“Uncovering the nucleus-chloroplast communication pathway in plants could yield new insights into mitochondrial gene expression in human cells and its misregulation in cancers,” Chen said.